Asymptomatic carriers drive E. coli spread in households, researchers find
The Yong Loo Lin School of Medicine, National University of Singapore (NUS Medicine), recently conducted a groundbreaking study on the transmission dynamics of Escherichia coli sequence type 131 (E. coli ST131) within households. This multidrug-resistant strain of E. coli has become a significant concern due to its resistance to antibiotics and its ability to cause serious infections.
The research, published in Nature Communications, revealed that some individuals can carry high levels of E. coli ST131 in their gut for extended periods without showing any symptoms. These “silent carriers” can unknowingly pass on the bacteria to their household members, contributing to the spread of the antibiotic-resistant strain within the community.
E. coli is a common bacterium found in the intestines of humans and animals. While most strains are harmless, some can cause infections when they acquire specific genes that make them pathogenic. E. coli ST131 is particularly concerning because it is resistant to multiple antibiotics, making infections difficult to treat.
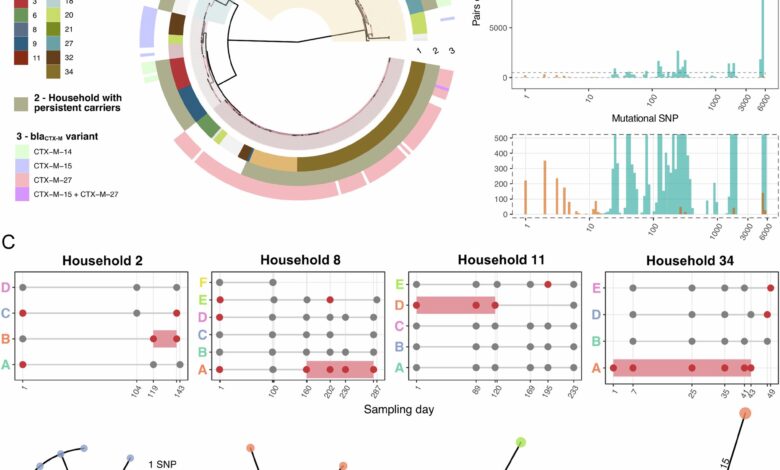
The study followed 34 families in Singapore, collecting stool samples from 135 participants to test for E. coli. Genetic sequencing was used to identify E. coli ST131 and track its spread within households. The researchers found that a small number of individuals carried the bacteria persistently and in high numbers, acting as potential sources of transmission to their family members.
Dr. Mo Yin, who led the study, emphasized the importance of identifying individuals who carry resistant bacteria without symptoms to implement targeted prevention strategies. The findings highlight the need for good personal hygiene practices within households and the development of new approaches to reduce the long-term carriage of antibiotic-resistant bacteria.
Potential strategies to combat the spread of E. coli ST131 include vaccines, probiotics, prebiotics, or fecal transplants. Targeting interventions at individuals who carry high levels of resistant bacteria could help reduce community transmission and combat antibiotic resistance.
Moving forward, the research team plans to further explore the gut microbiome of participants to understand how the balance between beneficial and harmful bacteria influences the long-term carriage of resistant strains. These insights will contribute to global efforts to address the rising threat of antibiotic resistance.
This study sheds light on the silent spread of antibiotic-resistant bacteria within households and underscores the importance of community-based solutions to contain antibiotic resistance. By understanding how these bacteria persist and move between individuals, researchers can develop practical strategies to mitigate the impact of superbugs like E. coli ST131 on public health.