Alito temporarily restores FDA rule allowing abortion pill mifepristone to be sent by mail
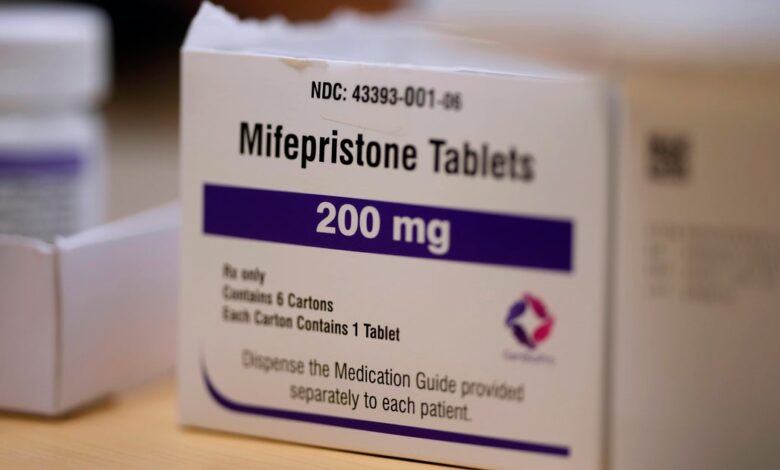
Supreme Court Justice Samuel Alito has temporarily halted an appellate court order that blocked an FDA rule allowing the abortion pill mifepristone to be prescribed online and dispensed through the mail. The decision comes after the makers of mifepristone, Danco Laboratories, and GenBioPro, the manufacturer of a generic version of the drug, requested relief from the Supreme Court.
Alito’s administrative stay will be in effect until May 11, giving the Supreme Court more time to consider the drug companies’ requests to set aside the appellate court’s order while the legal battle continues. Danco and GenBioPro have also asked the high court to take up the case and decide on the legal merits, with Louisiana officials given a deadline to respond to the drugmakers’ emergency appeals by Thursday.
This latest development brings the issue of mifepristone’s availability back to the Supreme Court for a second time. Since the overturning of Roe v. Wade in 2022, anti-abortion rights groups have been seeking to restrict access to the widely used abortion pill, claiming that the FDA did not adequately consider the drug’s safety and effectiveness when it was first approved in 2000.
In 2024, the Supreme Court unanimously rejected a challenge from a group of anti-abortion rights doctors and medical groups, ruling that they did not have the legal right to sue the FDA over mifepristone’s availability. This decision upheld a series of actions by the FDA since 2016 that made the abortion pill more accessible, including allowing it to be taken later in pregnancy and removing the in-person dispensing requirement.
The controversy over mifepristone escalated when the state of Louisiana filed a lawsuit challenging the FDA’s decision to allow the drug to be delivered by mail. Although a federal district court paused the litigation in April, the U.S. Court of Appeals for the 5th Circuit temporarily blocked the 2023 FDA rule that permitted healthcare providers to prescribe mifepristone remotely and send it through the mail.
The 5th Circuit panel found that Louisiana had legal standing to sue, citing concerns about the state’s Medicaid program covering emergency care for women who experienced complications after receiving the abortion pill from out-of-state providers. Louisiana’s laws protecting unborn human life were considered to be undermined by the 2023 regulation, according to the judges.
Danco and GenBioPro’s emergency appeals to the Supreme Court followed the 5th Circuit’s decision, with both companies expressing concerns about the immediate confusion and upheaval caused by the ruling. They argued that patients, providers, and pharmacies were left in limbo without proper guidance, potentially disrupting time-sensitive medical care for women across the country.
The White House has stated that they are closely monitoring the ongoing litigation on this issue, reaffirming their commitment to President Trump’s pro-life, pro-family, and pro-safety agenda. As the legal battle continues, the future of mifepristone’s availability and access remains uncertain, with implications for women’s reproductive rights and healthcare nationwide. The FDA is currently conducting a comprehensive and rigorous review of mifepristone, a medication used for medication abortion, in response to widespread concerns and controversies surrounding its safety and efficacy. This review is being conducted using the Gold Standard Science approach, which involves meticulous examination and analysis of all available scientific data and evidence.
The FDA’s primary goal in this review is to address the various concerns and uncertainties that have been raised regarding the use of mifepristone. Despite the medication being approved for use in medication abortion for several years, there have been ongoing debates and discussions about its safety, potential risks, and long-term effects.
The FDA’s commitment to conducting a thorough review is aimed at providing clarity and reassurance to both healthcare providers and patients who may be considering or currently using mifepristone. By adhering to the Gold Standard Science approach, the FDA is ensuring that all aspects of the medication, including its safety profile, effectiveness, and potential side effects, are carefully evaluated and analyzed.
This review process is essential in addressing any gaps in existing knowledge and understanding of mifepristone, as well as in addressing any emerging concerns or issues that may have arisen since its approval. By conducting a comprehensive review, the FDA is working to ensure that the medication continues to meet the highest standards of safety and effectiveness.
In the meantime, healthcare providers and patients are encouraged to stay informed and updated on the FDA’s review process and any developments related to mifepristone. It is important for individuals to consult with their healthcare providers and seek accurate and reliable information when making decisions about their reproductive health.
Overall, the FDA’s ongoing review of mifepristone underscores the agency’s commitment to upholding the highest standards of scientific rigor and ensuring the safety and well-being of patients. By conducting a thorough and comprehensive review, the FDA is working to address concerns and provide clarity on the use of mifepristone in medication abortion.





