Bird flu found to evolve rapidly, infecting more hosts and finding ways to spread more effectively
Avian flu, also known as H5N1, has been quietly evolving in the background, aiming to become more effective and widespread. Since its detection in Canada in 2021, the highly pathogenic avian flu has infected over 180 million domestic birds in both Canada and the United States. A recent study published in Science Advances reveals that the H5N1 virus has been mixing with low-pathogenicity avian influenza viruses (LPAIVs) to broaden its host range and geographical distribution.
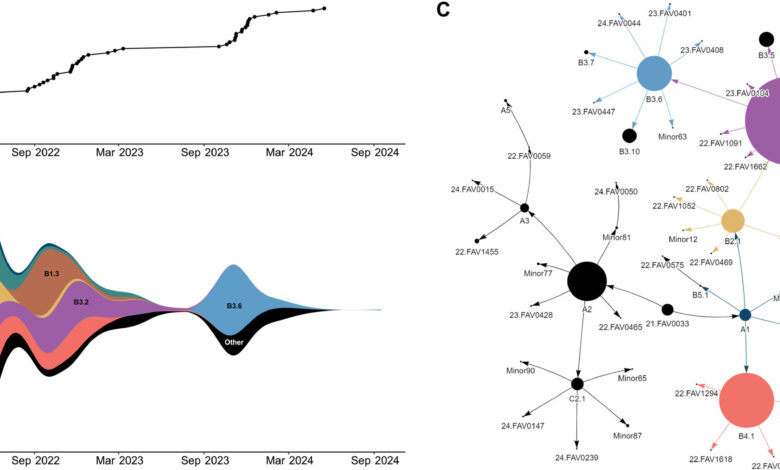
Researchers conducted an analysis of 2,955 viral genomes collected from infected domestic and wild birds in Canada between 2021 and 2024. By combining this data with previously published analyses, they were able to track the genetic evolution and geographic spread of the bird flu virus across North and South America, largely facilitated by migratory birds.
The study uncovered a significant increase in the viral fitness of the bird flu virus, attributed to frequent reassortment events. Reassortment occurs when two or more viruses infect the same cell, leading to the creation of a new virus with a unique combination of genetic characteristics. This process often results in a virus with enhanced ability to infect a wider range of hosts and spread more effectively. The study indicates multiple reassortment events within the bird flu virus, with newer genotypes outcompeting previous ones.
Certain regions, such as the Prairie Pothole Region in the Great Plains, serve as hotspots for viral transmission due to the high concentration of migratory birds passing through the area. The region, known for its abundant waterfowl population, provides ample opportunities for different avian flu strains to intermingle and generate stronger variants.
The study found that new genotypes and peaks in viral detection coincided with migratory periods in May 2022, October 2022, and October 2023, suggesting that wild birds are the primary carriers of the newer strains. This shift from primarily circulating in domestic poultry to wild birds poses a challenge in disease containment due to migration patterns. Additionally, evidence of transmission to mammals, which then spread back to birds, was observed.
Given the high mortality rate in poultry and the potential spread to other animals and humans, it is crucial to prioritize efforts in mitigating bird flu. Further research is essential to understand viral evolution and develop strategies for disease control in both poultry industries and wildlife management. By gaining a deeper understanding of viral dynamics, we can better prevent future pandemics and safeguard against the development of more virulent strains.
This article was written by Krystal Kasal, edited by Gaby Clark, and fact-checked by Andrew Zinin. For more information, refer to the study published in Science Advances titled “Spatiotemporal reconstruction of the North American A(H5N1) outbreak reveals successive lineage replacements by descendant reassortants” (DOI: 10.1126/sciadv.adu4909).





