FDA approves first new gonorrhea drug in decades
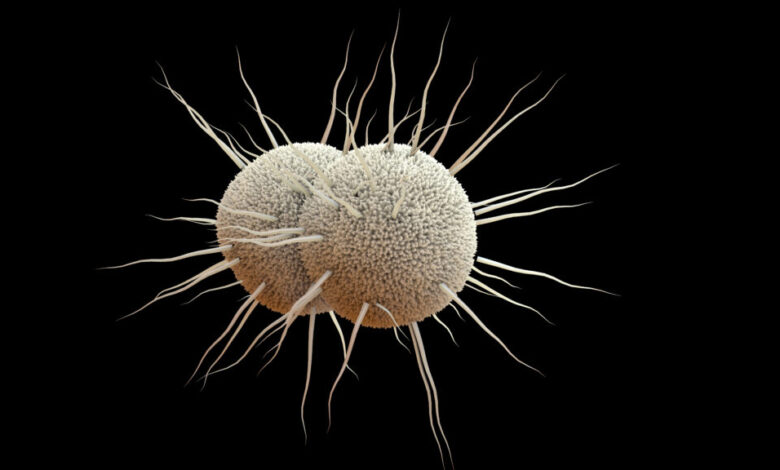
The battle against the antibiotic-resistant bacterium that causes gonorrhea has taken a positive turn with the recent approval of GSK’s Blujepa by the Food and Drug Administration. Blujepa, also known as gepotidacin, is a new class of antibiotic that has been granted supplemental approval for the treatment of uncomplicated urogenital gonorrhea. This approval comes as a much-needed relief as the bacterium Neisseria gonorrhoeae has been steadily developing resistance to all available antibiotics, leaving only ceftriaxone as a last resort.
This groundbreaking approval marks the first new class of antibiotics for gonorrhea in over three decades. The drug was initially approved earlier this year for the treatment of uncomplicated urinary tract infections in women and adolescents aged 12 and older. GSK’s chief scientific officer, Tony Wood, expressed pride in delivering this new treatment option, emphasizing the importance of expanding the range of effective oral treatments for gonorrhea.
Renowned clinician and researcher specializing in sexually transmitted infections, Jeanne Marrazzo, hailed the approval of Blujepa as a significant milestone in combating the challenges posed by antibiotic-resistant gonorrhea. Marrazzo highlighted the ongoing struggle in managing this pathogen’s capacity for developing resistance and welcomed the availability of an effective and safe first-in-class antibiotic.
Blujepa’s oral administration is a notable advantage over ceftriaxone, which requires intramuscular injection. However, Blujepa must be administered in two doses. The approval specifies that the drug is intended for use in adult and pediatric patients aged 12 and above, weighing at least 45 kilograms, who have limited or no alternative treatment options for uncomplicated urogenital gonorrhea.
Gonorrhea, the second most commonly diagnosed sexually transmitted infection in the US, has a notorious history of developing resistance to antibiotics, earning it priority pathogen status from the World Health Organization and a classification as an urgent public health threat by the CDC. Untreated gonorrhea can lead to infertility and other serious sexual and reproductive health complications.
In addition to Blujepa, another potential treatment option for gonorrhea is on the horizon. Zoliflodacin, a first-in-class antibiotic developed by Innoviva Specialty Therapeutics and GARDP, is awaiting FDA approval with a decision date set for December 15. The emergence of these new treatment options brings hope in the ongoing battle against antibiotic-resistant gonorrhea, offering much-needed alternatives to combat this persistent public health threat.





