New hepatitis B vaccine guidelines seem likely to be ignored
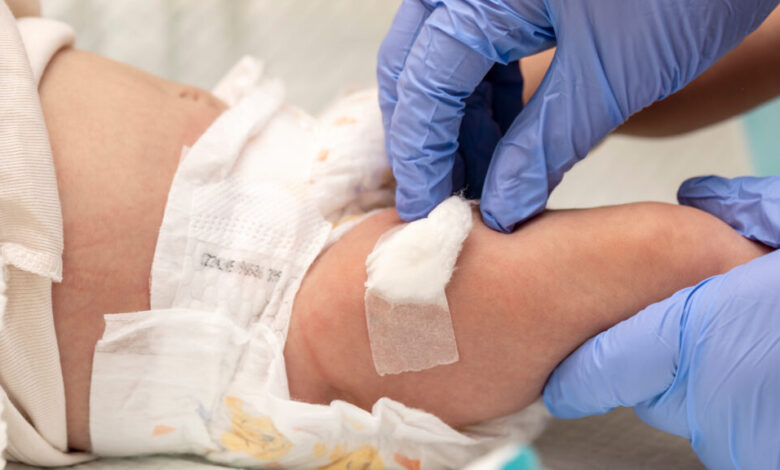
The decision by the Trump administration to adopt the vaccine advisory committee’s recommendation to end the policy of offering every newborn a hepatitis B shot has sparked controversy among health experts. Despite the outcry, the Centers for Disease Control and Prevention (CDC) now advises parents to discuss with their doctors whether to vaccinate infants at birth against hepatitis B, specifically for babies born to mothers who test negative for the virus.
However, some doctors and hospital systems have chosen to continue following the previous guidance from the American Academy of Pediatrics or their state health departments, rather than the new federal recommendation. This divergence in practice reflects the increasing fragmentation of U.S. vaccine policy under the current administration.
Dr. Vandana Madhavan of Massachusetts General Hospital expressed concern about the lack of evidence supporting the new CDC recommendation, emphasizing the importance of evidence-based guidelines that have been established for decades. The CDC’s decision to change the policy for infants born to hepatitis B-negative mothers has raised questions and resistance among healthcare providers.
While some health systems have pledged to continue offering the hepatitis B shot at birth, others have expressed reservations about the new guidance. Kaiser Permanente, Children’s National, and Denver Health are among those maintaining the current practice of vaccinating newborns against hepatitis B. The resistance from healthcare providers poses a challenge to officials seeking to implement nationwide changes in vaccination practices.
Despite the CDC’s assertion that the recommendation reflects a rigorous review of available evidence, some state health departments and public health groups have reaffirmed their support for the birth dose of the vaccine. The debate over the new CDC recommendation underscores the complexities of aligning vaccine policies and practices at a national level.
In response to the resistance from healthcare providers, some of the administration’s vaccine advisers have criticized the reluctance to adopt the new guidance. ACIP’s vice chair, Robert Malone, attributed the resistance to self-interest within the vaccine industry, while ACIP member Retsef Levi emphasized the importance of evidence-based science in restoring public trust.
As the committee continues its review of the childhood vaccine schedule, the mixed responses from healthcare providers suggest that the impact of the hepatitis B recommendation may not be as significant as initially anticipated. The debate surrounding the CDC’s decision highlights the challenges of balancing evidence-based science with individual healthcare practices.





