Vertex’s CRISPR sickle cell treatment faces a costly bottleneck
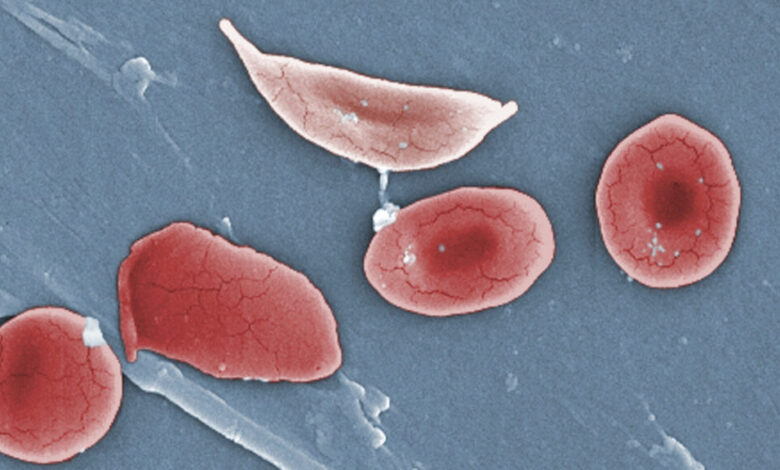
Vertex executives had warned that the rollout of Casgevy, their groundbreaking curative treatment for sickle cell disease, would be slow. However, the pace at which the therapy is reaching patients has surprised many in the medical community.
Despite being approved over two years ago, only around 60 patients in the United States, Middle East, and Europe have received treatment with this gene-editing therapy. Specialists at leading sickle cell centers have identified a major obstacle hindering the wider distribution of Casgevy: the inability to collect enough cells to produce the treatment.
This challenge couldn’t have come at a more crucial time for Vertex, as the company is striving to establish Casgevy in the market while also facing competition from rival therapies. Furthermore, a significant competitor is expected to enter the scene next year, intensifying the pressure on Vertex to accelerate the delivery of their treatment to patients in need.
The slow progress in the distribution of Casgevy highlights the complexities involved in bringing cutting-edge therapies to the market and delivering them to the patients who can benefit from them. Vertex’s efforts to overcome the cell collection bottleneck and expand access to Casgevy will be closely watched by the medical community and patients eagerly awaiting this potentially life-changing treatment.





