Fulcrum sickle cell pill data at ASH: Higher dose works better
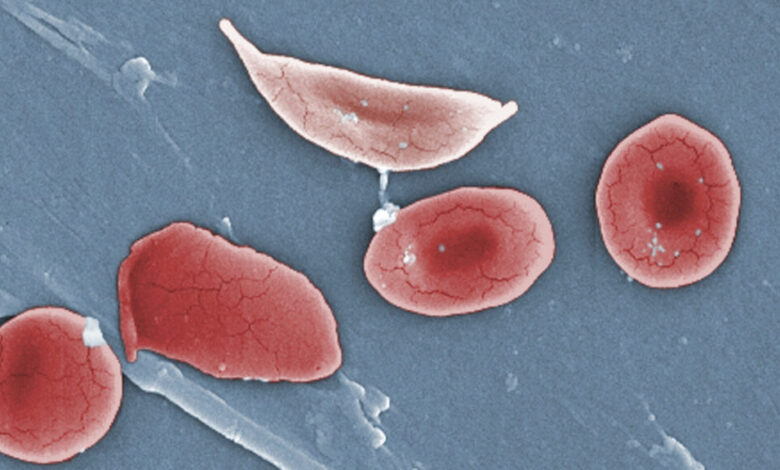
Fulcrum Therapeutics announced on Sunday that a higher dosage of its experimental medication for sickle cell disease showed promising results in inducing fetal hemoglobin, a different form of the oxygen-carrying protein hemoglobin. This breakthrough has raised hopes for a potential simple and effective treatment for the debilitating disease.
Sickle cell disease is a genetic condition caused by a mutation in the hemoglobin gene, resulting in misshapen blood cells that obstruct blood vessels and hinder the proper delivery of oxygen throughout the body. The medication, known as pociredir, is specifically designed to stimulate the production of fetal hemoglobin, a type of hemoglobin that is typically only active during fetal development and infancy.
In a recent early-stage study presented at the American Society of Hematology annual meeting, 12 participants who were administered a daily dose of 20mg of pociredir experienced a significant increase in their levels of fetal hemoglobin. Prior to the treatment, the participants had fetal hemoglobin levels accounting for 7.1% of their total hemoglobin, which rose to 16.9% after receiving the medication.
The findings from this study are promising as they demonstrate the potential of pociredir to effectively boost fetal hemoglobin levels in individuals with sickle cell disease. By increasing the presence of fetal hemoglobin, the medication has the potential to alleviate the symptoms and complications associated with the disease, offering hope for a better quality of life for patients.
Overall, the development of pociredir and its ability to induce fetal hemoglobin represent a significant advancement in the treatment of sickle cell disease. With further research and clinical trials, this medication could potentially become a game-changer in the management of the condition, providing patients with a much-needed treatment option that is both effective and accessible.